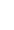Initial Treatment Decisions
Patients who receive a new breast cancer diagnosis are understandably anxious to find out as soon as possible what treatment will entail. It is rarely possible at the time of the initial diagnosis to accurately predict exactly what treatment will be required in their particular situation, but often with the information obtained from the clinical breast examination, the initial breast imaging and the needle core biopsy pathology, it is usually possible for some preliminary predictions to be made.
As the breast surgeon is usually the first specialist with whom the patient meets, part of my role is to explain the overall treatment process, not just the surgical component, and the rationale for considering other forms of treatment, such as drug therapy and radiotherapy, in addition to surgery.
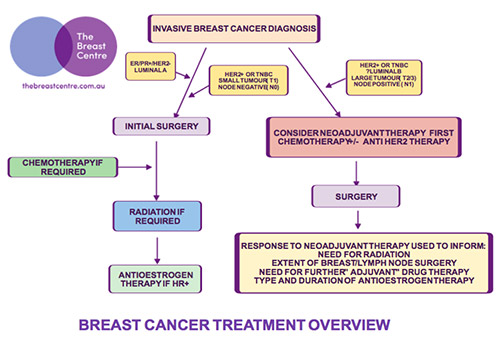
The management of breast cancer is a step by step process, with the outcome of each stage often determining what is required next, and as such, I find a helpful way of outlining the possible treatment pathways is with the use of treatment algorithms, which outline the decision making process in a flow chart format.
Practices change over time, with the introduction of modern surgical techniques and new drug therapies, and I have developed my own breast cancer treatment algorithms, based on the way I currently work through the decision making in each individual patient. See Breast Cancer Treatment Algorithms
The management of early breast cancer is based on “tumour burden” and “molecular subtype”. The two major pillars of breast cancer treatment are “locoregional” and “systemic” treatment. Locoregional therapy refers to the local treatment of the breast and axilla, with surgery +/- local radiotherapy.
The term “systemic “therapy refers to all types of drug therapy, including chemotherapy, biologic targeted therapy, and endocrine therapy. “Adjuvant” (post-operative) systemic therapy is administered in addition to local therapy to treat any potential “micrometastatic” disease, and thus hopefully prevent distant recurrence, and is tailored to the cancer subtype and risk of recurrence. “Neoadjuvant” (preoperative) systemic therapy targets local disease (breast/axilla) as well as potential micrometastatic disease elsewhere in the body.
Once a diagnosis of breast cancer is established, the extent of local disease is assessed to determine whether the tumour is “operable” or “inoperable”.
The clinical criteria of “inoperability” include inflammatory carcinoma, fixation of the tumour to the bony chest wall (ribs, sternum), extensive skin involvement with ulceration or satellite skin nodules, fixed/ matted axillary lymphadenopathy, involvement of neurovascular structures of the axilla, or lymphoedema (swelling) of the arm. All of these findings are identifiable on physical examination, and if present should prompt imaging evaluation for distant metastases. “Inoperable” breast cancer in the absence of distant metastatic disease, is a type of “locally advanced breast cancer (LABC), and is uncommon.” In these cases, systemic (drug) therapy is administered as the initial treatment in order to reduce tumour volume, the aim being to render the tumour “operable”.
Preoperative systemic drug therapy most commonly takes the form of chemotherapy, which is termed “Neoadjuvant” chemotherapy (NAC). In HER2 +ve breast cancer, anti-HER2 agents, either single or dual, are also given preoperatively in conjunction with chemotherapy. Although preoperative oral anti-hormonal drug therapy is an option for hormone receptor +ve/HER2 -ve breast cancers, partly due to the slow rate of clinical response, it is uncommonly considered, except on occasions in the older breast cancer patient.
NAC was initially utilized as a way of rendering locally advanced, inoperable breast cancer resectable. More recently however NAC has been increasingly used in operable tumours to downstage disease in the breast and axilla, with the intention of facilitating breast conservation and, in some instances, avoiding axillary lymph node dissection (ALND). The oncologic safety and equivalent survival outcomes of NAC have been demonstrated in several randomized trials, and a meta-analysis of patients treated with NAC versus surgery followed by chemotherapy has similarly shown no difference in survival.
The vast majority of women have “operable” breast cancer at the time of diagnosis, and in this situation the sequencing of surgical resection and systemic therapy is variable. Depending on the tumour subtype and the local extent of disease in the breast and/or axilla, preoperative systemic therapy may be used to reduce tumour volume. Preoperative drug therapy importantly also enables the assessment of the tumour’s responsiveness to drug treatment, as evidenced by operative pathology, which evaluates the level of response, according to whether there is residual tumour present, and if so, the volume of residual disease.
THE FIRST STEP:
When I see a patient with a new operable invasive breast cancer diagnosis, there are three important pieces of information in which I am initially most interested, because they are crucial in informing the nature and sequencing of the initial breast cancer treatment.
These three factors are:
- “Molecular subtype” of the cancer on needle core biopsy
- Tumour Size
- Axillary Nodal Status
-
Molecular Subtype
In days gone by, the only information usually provided by the diagnostic needle biopsy was the confirmation of the breast cancer diagnosis. Pathology has advanced significantly in recent years, and the level of information regarding the biological nature of the cancer that we currently obtain from the core biopsy pathology report was previously only available (if it all) on the surgical excision specimen. This information not only potentially influences the sequencing of treatment, but also enables the breast surgeon to counsel the patient at the time of diagnosis about the likelihood that chemotherapy, targeted agents and/or hormonal blockade will form part of their treatment, either prior to or after surgical removal of the cancer.
The pathological information that may be obtained from the diagnostic breast core biopsy includes:
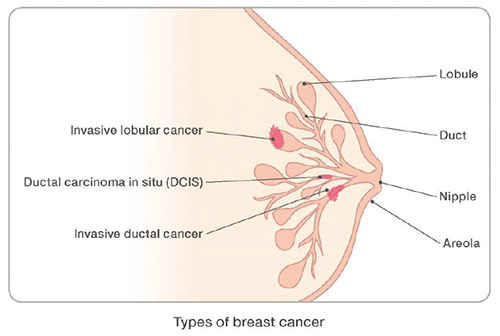
- Histological Type: The two most frequent histological types of breast cancer are invasive ductal cancer-often described as being of “no special type” (NST)- (70-75%) and invasive lobular cancer (ILC)-(10-15%). The other 18 subtypes are uncommon (0.5-5%). See My Pathology Report
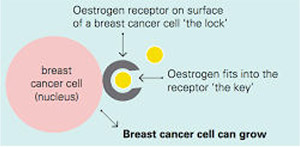
- Receptor Status: Oestrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2). See My Pathology Report
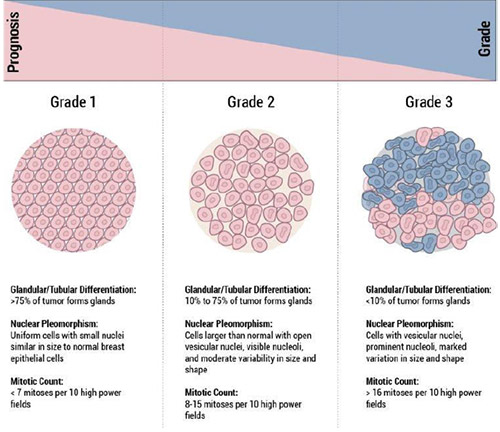
- Grade: The grade of a breast cancer indicates the pattern of the cancer cell growth and how fast the cancer cells are growing. The grade is numbered from 1 to 3:
- A provisional or estimated tumour grade is often reported on the core biopsy, but the final grade of the tumour can only be determined on the operative specimen. See My Pathology Report
- Grade 1 (Low Grade or Well Differentiated) The cancer cells still look a lot like normal cells. They are usually slow growing and are likely to behave less aggressively.
- Grade 2 (Intermediate/Moderate Grade or Moderately Differentiated) The cancer cells do not look like normal cells, and are growing somewhat faster than normal cells.
- Grade 3 (High Grade or Poorly Differentiated) The cancer cells do not look at all like normal cells. This means the cells are growing more quickly and are more likely to be aggressive.
+/- Ki 67: Ki-67 is a protein in cells that increases when cells are dividing, and is a marker of proliferation. It is reported as the percentage of cancer cells that contain Ki-67.The more positive cells there are, the more quickly the cancer is dividing and growing.
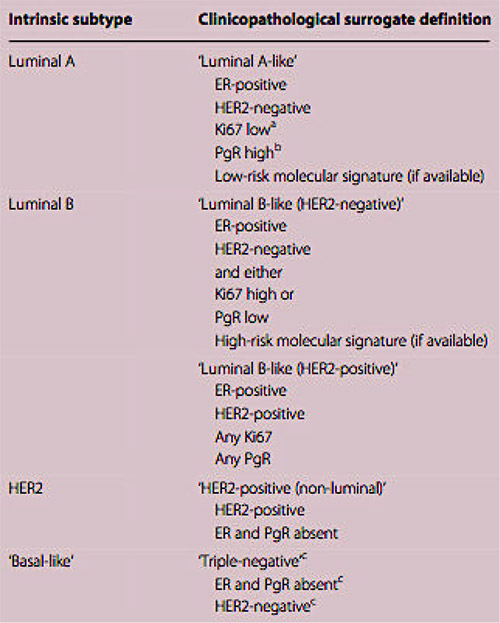
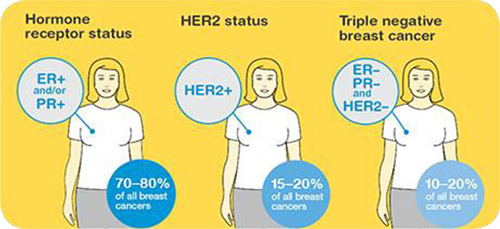
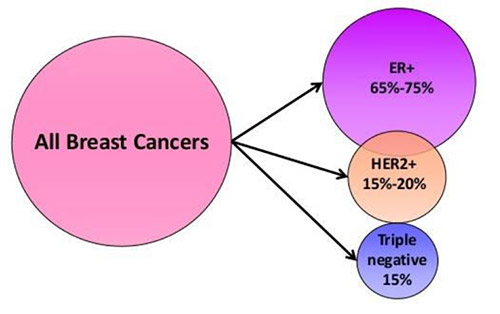
Clinical practice typically uses a classification of five molecular breast cancer subtypes based on the histology and immunohistochemistry expression of key proteins: oestrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) and the proliferation marker Ki67. A provisional estimate of the tumour grade is also usually provided. Tumours expressing ER and/or PR are termed ‘hormone receptor-positive’ (HR+) breast cancers , those expressing human epidermal growth factor receptor 2 are described as HER2 positive (HER2+) and can be hormone receptor positive or negative. Tumours not expressing ER, PR or HER2, are called ‘triple-negative’ breast cancers (TNBC).

If the needle core biopsy confirms the tumour to be either HER2+ or triple negative, it is almost inevitable that there will be a recommendation for chemotherapy to be administered at some point, either prior to, or after surgery, and in HER2 + disease, anti-HER2 therapy in addition to chemotherapy. When it appears inevitable that chemotherapy will be recommended, based on the tumour biology/molecular subtype on the initial diagnostic core biopsy, I think it is appropriate to advise patients of this early on, as amongst other things it serves to reinforce the concept that the recommendation for chemotherapy is largely made on the basis that a tumour is highly likely to be “chemosensitive”, based on tumour biology, and that the requirement for chemotherapy does not necessarily reflect a more advanced cancer or a worse prognosis, but that chemotherapy is the most appropriate drug therapy in that molecular subtype of breast cancer.
- HR+/HER2- tumours form the most common subtype, and endocrine therapy to block ER activity is recommended all patients with ER+ and/or PR + breast cancers. Whether chemotherapy is required in HR+/HER2- tumours can often only be determined after operative pathology is available, sometimes with the additional assistance of a genomic test such as Oncotype DX. Oncotype DX analyses 21 genes within a sample of tumour tissue to predict the risk that the breast cancer may recur. It may be helpful in determining whether or not you would bene t from chemotherapy. If the test shows you have a higher risk of recurrence, chemotherapy may be recommended. If your risk of recurrence is low, you may be able to avoid chemotherapy and be treated with anti-hormonal therapy alone.Invasive lobular carcinomata (ILC) are almost always strongly HR+ and HER2-. Preoperative MRI is commonly undertaken in ILC.
For the purposes of clinical decision making, I find when discussing breast cancer subtypes with patients, that it is helpful to consider 4 main categories, as in the table below, which I’m sure many of my patients will recognize.

Breast cancer core biopsies that report receptor status allow tumours to be divided into four categories, which in turn enables me to immediately develop and discuss with the patient a preliminary treatment “template”. On occasions, the core biopsy HER2 result is equivocal, and the final HER2 status of the tumour may sometimes not be known until the definitive test is performed. This is usually undertaken on the operative specimen, but if preoperative drug therapy is being considered, it can be performed on the core biopsy specimen.
For cancers which fall into categories 2, 3 and 4 in the table above, there will almost always be a recommendation for chemotherapy +/- HER2 targeted therapy at some point. What sort of chemotherapy, for how long and whether it is administered before or after surgery is influenced by the “tumour burden”, which is determined by the “size” and “nodal status” of the cancer, not the biological “molecular subtype”. The tumour biological subtype determines what class of drug therapy is appropriate, and the size and nodal status allow that decision making to be further refined. In general terms, as chemotherapy attacks rapidly dividing cells, it tends to be most effective in faster growing cancers, which are typically high grade and HR-, and these cancers are usually what we describe as “chemosensitive”.
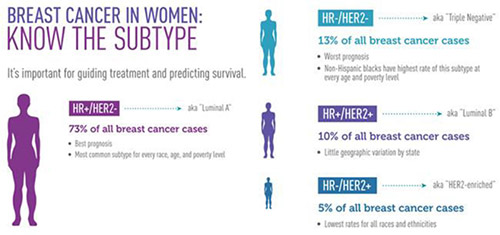
Cancers which fall into category 1, which are incidentally by far the most common, do not automatically prompt a recommendation for chemotherapy. As often there may not even be a need for chemotherapy in these cancers, preoperative chemo is therefore not commonly considered, and the majority of category 1 tumours (HR+/HER2-) are treated with initial surgery. Decisions regarding postoperative (adjuvant) systemic (drug) therapy are then made after surgery, based on the operative pathology results. As outlined above, endocrine therapy is recommended at some point for all HR+ breast cancers.
Some Category 1 cancers appear more proliferative in nature on the preoperative needle core biopsy, as evidenced by high grade, low or absent progesterone receptor staining and an elevated KI67, and may be described as “luminal B”. These tumours can often prove to be chemosensitive, and preoperative chemo may be considered on occasions, particularly if spread to the nodes has been pathologically confirmed preoperatively by needle biopsy. Low grade tumours which are strongly ER and PR +, with a low Ki67, known as “luminal A” tumours, tend as a group to be fairly slow growing, and may not derive significant benefit from chemotherapy even if large and node positive, as the tumour is itself relatively “chemoresistant”.
- Histological Type: The two most frequent histological types of breast cancer are invasive ductal cancer-often described as being of “no special type” (NST)- (70-75%) and invasive lobular cancer (ILC)-(10-15%). The other 18 subtypes are uncommon (0.5-5%). See My Pathology Report
-
Tumour size
As assessed by:
- Clinical examination (if there is a palpable “lump”)
- Mammography- bilateral, preferably 3D digital mammography with tomosynthesis
- Ultrasound- formal ultrasound performed by the radiologist, sometimes supplemented by bedside ultrasound performed by the breast surgeon to correlate clinical and imaging findings.
- +/- MRI- see Diagnosis and the Triple Test
The estimated size of the tumour, as assessed by some or all of the modalities above, is important in helping to determine the most appropriate form of surgery to the breast. See: Lumpectomy vs Mastectomy: How to Choose

-
Axillary Nodal Status
As assessed by:
- Clinical examination- low sensitivity, other than the heavily involved axilla
- Targeted ultrasound-ultrasound guided needle biopsy, either fine needle aspirate (FNA) or preferably core biopsy, of any axillary nodes on the side of the breast cancer that are even marginally abnormal on ultrasound.
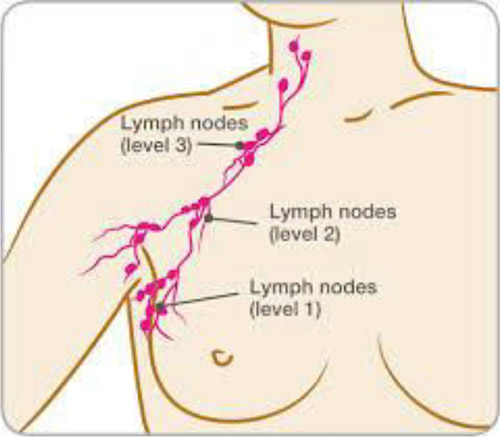
In patients with invasive breast cancer, other than those confirmed to be node +ve preoperatively, axillary staging takes the form of sentinel node biopsy. (SLNB). See Sentinel Node Biopsy. Currently, in patients with pathologically confirmed nodal involvement on preoperative image guided needle biopsy, who are undergoing surgery as the initial treatment, axillary node clearance (ANC) is usually undertaken. Node positive patients who undergo preoperative / Neoadjuvant chemotherapy may be eligible for staging of the axilla with more conservative surgery following treatment.
Armed with information on the tumour molecular subtype, size and nodal status, it is possible to commence treatment decision making.
Surgical Management of Breast Cancer
Understanding Breast Cancer
Breast Cancer
Breast Cancer Diagnosis and Treatment
How Surgery is used to treat Breast Cancer
Error: Contact form not found.